Next-Gen Precision Imaging, Navigation, & Robotics
Unlocking better outcomes, reducing system costs, and enabling the AI-driven operating room of tomorrow.
Mission
Our mission is to help clinicians see with greater precision and clarity, improving patient care everywhere.
Proven Innovation, Global Impact
A record of advancing transformative technologies, surpassing legacy systems, and setting a new standard for surgical excellence.
0+
Patient procedures
0 Countries
Deployed across
0+ Issued Patents
Across surgical robotics and optics, AI planning, navigation, and breakthrough MRI
Proven Leap in Technology
Against legacy microscopes and MRIs
Analog Tools Can’t Meet Modern Clinical Demands
Outdated imaging and visualization systems limit data, fragment workflows, and prolong treatment - leading to poorer outcomes and clinician frustration. It’s time to move beyond legacy technologies that hold hospitals back.
why synaptive
The Synaptive Advantage
Better Outcomes
Faster, safer, more precise surgeries
Lower Costs
Reduced OR time and shorter hospital stays
Breakthrough Technology
Designed for AI-powered surgical care
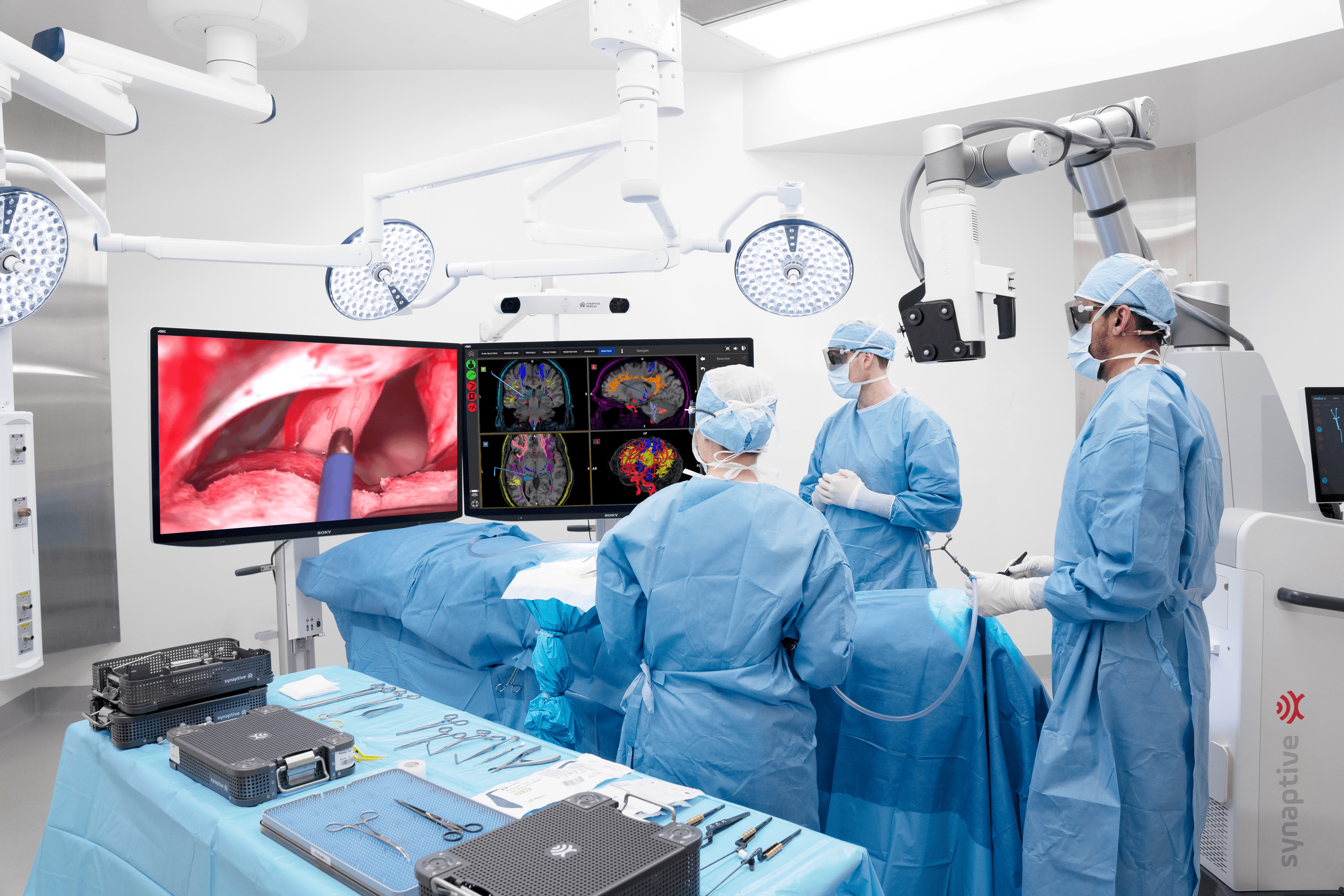
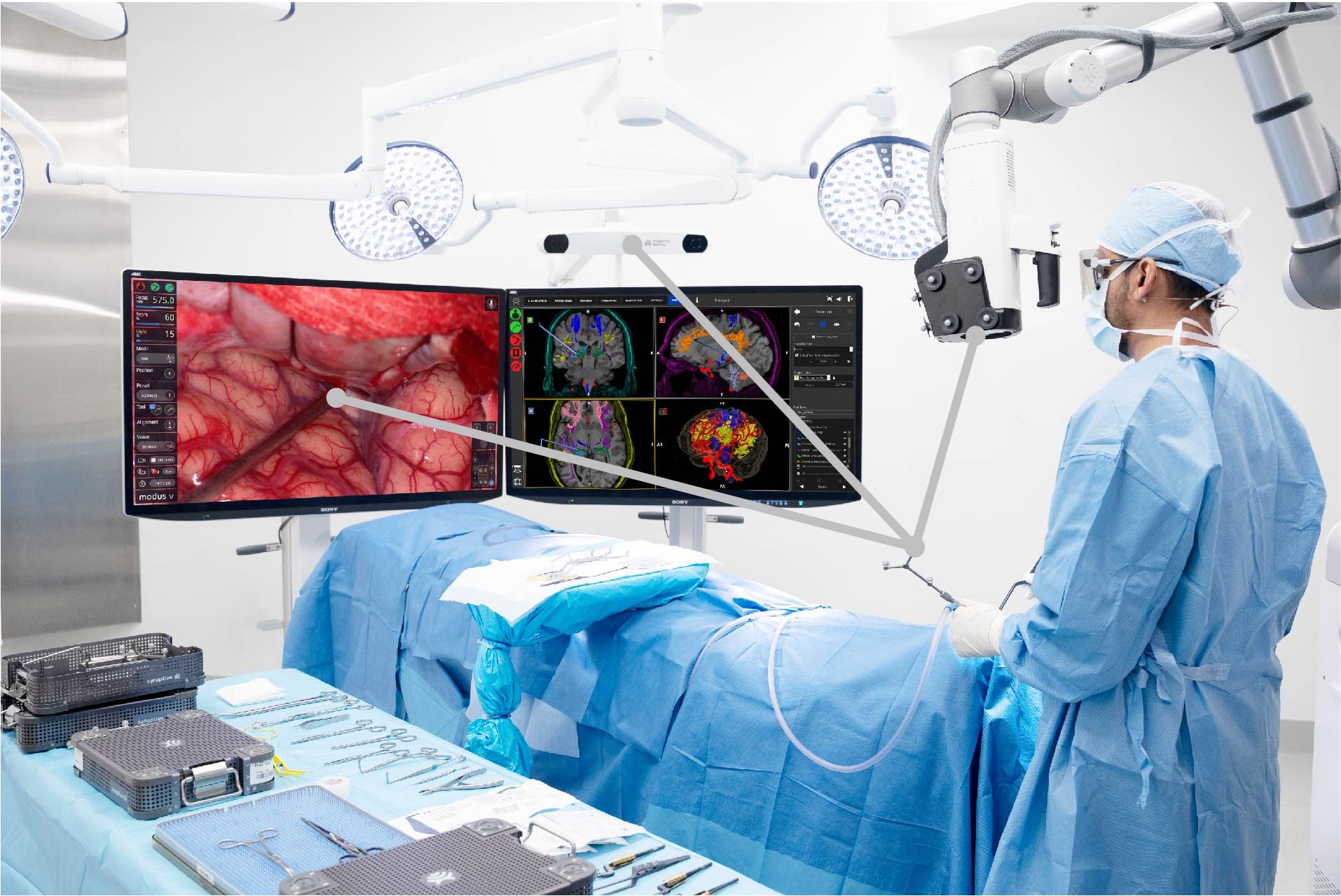

SYNAPTIVE INTEGRATED SUITE
Four Unique Products, One Transformational Platform
SYNAPTIVE INTEGRATED SUITE
Four Unique Products, One Transformational Platform
Integrated
Suite
Suite
DIAGNOSIS
REVIEW
Treatment
MANAGEMENT
EVRY MRI
Next-generation, low-field 0.5T MRI with best-in-class head imaging.
Modus Plan
Automatic tractography segmentation accessing patient-specific fiber bundles in seconds.
Modus X
Robotic exoscope featuring advanced 4K 3D optics and fluorescence visualization.
Modus Nav
Enhanced navigation with best-in-class tractography for safer, more effective neurosurgical procedures.
Diagnosis
Review
Treatment
Management





CASE STUDY: Stephen's Story
“Stephen’s tumor was in an especially intricate area of the brain. On top of that, he also had epilepsy, with seizures triggered by irritation around the tumor. For his surgery, we used the Synaptive Modus Exoscope, which lets you see details and angles that were never possible before. The surgery was successful, and today Stephen is doing great and is seizure-free.”

Dr. Ramin Eskandari
Medical University of South Carolina
0x
Systems purchased
0
Surgeon users
0
Applicable DRG’s
0%
Reduction in operative time
0%
Reduction in length of stay
Elite Clinical Adoption
Trusted by the Best
Adopted by 100+ top centers worldwide, including 6 of the top 10 U.S. neurosurgery hospitals.










.png)











